Supakul’s Group

Research Overview
We aim to advance understanding of psychiatric and neurological disorders and support drug discovery by generating patient-derived iPS cells and differentiating them into diverse neural cell types in vitro for comprehensive analysis.
1. Establishment of Patient-Derived Human iPS Cells
In collaboration with clinical departments, we collect patient samples—such as blood or urine—and process them into culture-compatible cells. These cells are reprogrammed into iPS cells, generating original patient-specific iPS cell lines.
The established iPS cells undergo rigorous quality control, including karyotype analysis, assessment of pluripotency marker expression, and verification of trilineage differentiation potential (Fig. 1). Once validated, these iPS cell lines are used for differentiation into various neural cell models.
 Figure 1. Derivation and Quality Evaluation of Human iPS Cells
Figure 1. Derivation and Quality Evaluation of Human iPS Cells
References
Sopak Supakul, Hideyuki Okano. NEUROLOGICA. 2022
2. Development of Co-Culture Systems Using Human iPS Cell–Derived Neural Cells and Their Application to Neurodegenerative Disease Research
Using established iPS cell lines, we differentiate pluripotent cells into various neural cell types through multiple induction protocols. We then combine these neurons with astrocytes to develop co-culture systems. Such systems enhance cellular maturity, promote in vivo-like morphology in each cell type, and enable interactions such as tripartite synapse formation and paracrine signaling.
These features make the co-culture system a powerful tool for disease modeling and drug discovery research using human cells, particularly for neurodegenerative diseases (Fig. 2).
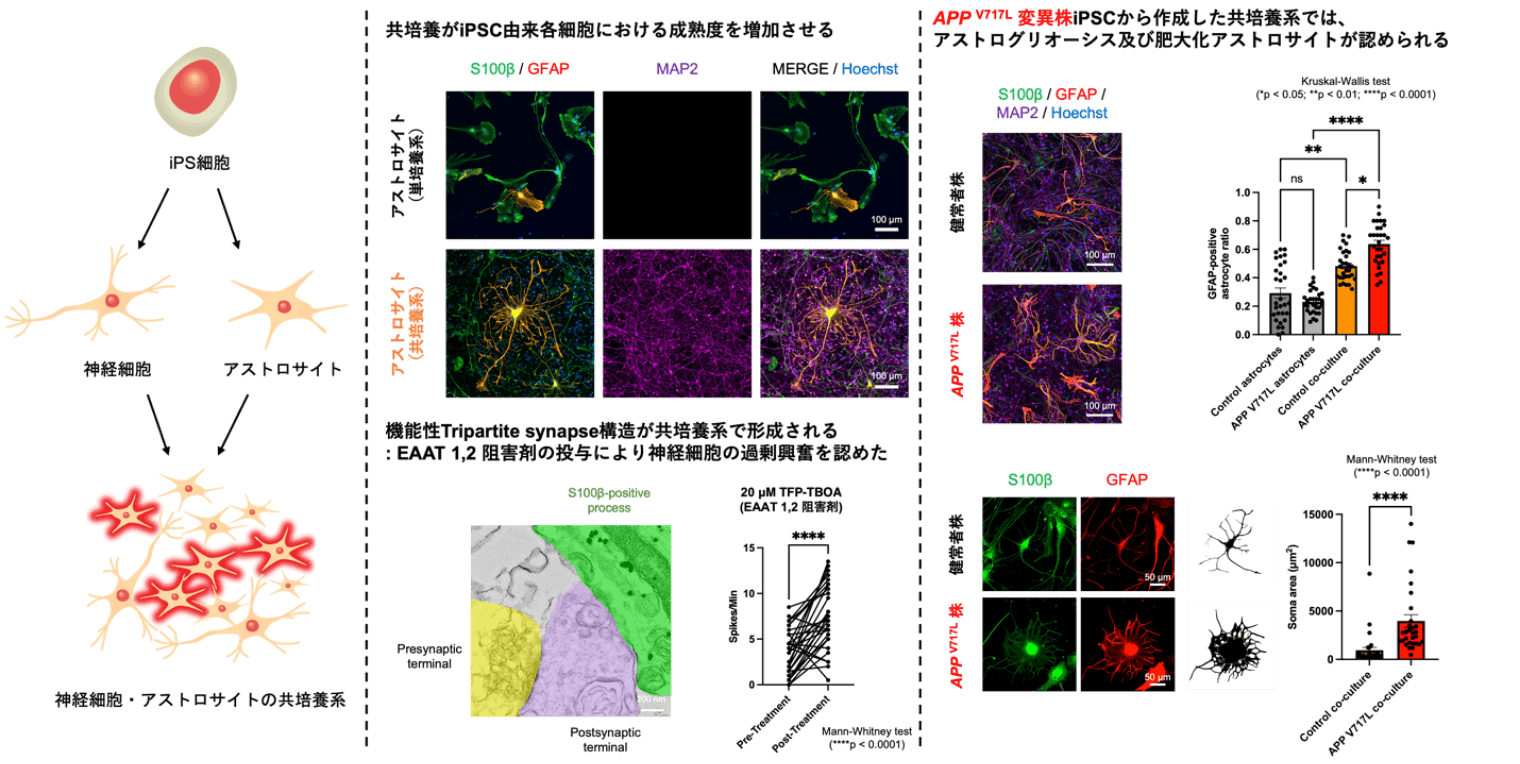 Figure 2. Development of a Co-Culture System Using Human iPS Cell–Derived Neurons and Astrocytes, and Its Application to Phenotypic Analysis in Alzheimer’s Disease
Figure 2. Development of a Co-Culture System Using Human iPS Cell–Derived Neurons and Astrocytes, and Its Application to Phenotypic Analysis in Alzheimer’s Disease
References
3. Translational Research on Psychiatric Disorders Using Patient-Derived iPS Cells
In collaboration with the Department of Psychiatry at Fujita Health University, we are conducting studies to better understand the stratification of schizophrenia patients based on antipsychotic treatment response, as well as the mechanisms of action of these drugs. Specifically, we establish new iPS cell lines from patient blood samples and have developed a novel induction method for medium spiny neurons (MSNs)—the neuronal subtype expressing dopamine D2 receptors, the primary target of antipsychotics (Fig. 3).
Going forward, we aim to induce MSNs from patient-derived iPS cells to elucidate the mechanisms of dopamine D2 receptor antagonists from the perspective of treatment response. This work is expected to contribute to the development of new therapeutics for schizophrenia. Furthermore, by leveraging our current research platform, we anticipate expanding into the pathophysiological analysis and therapeutic development for a broad range of psychiatric disorders.
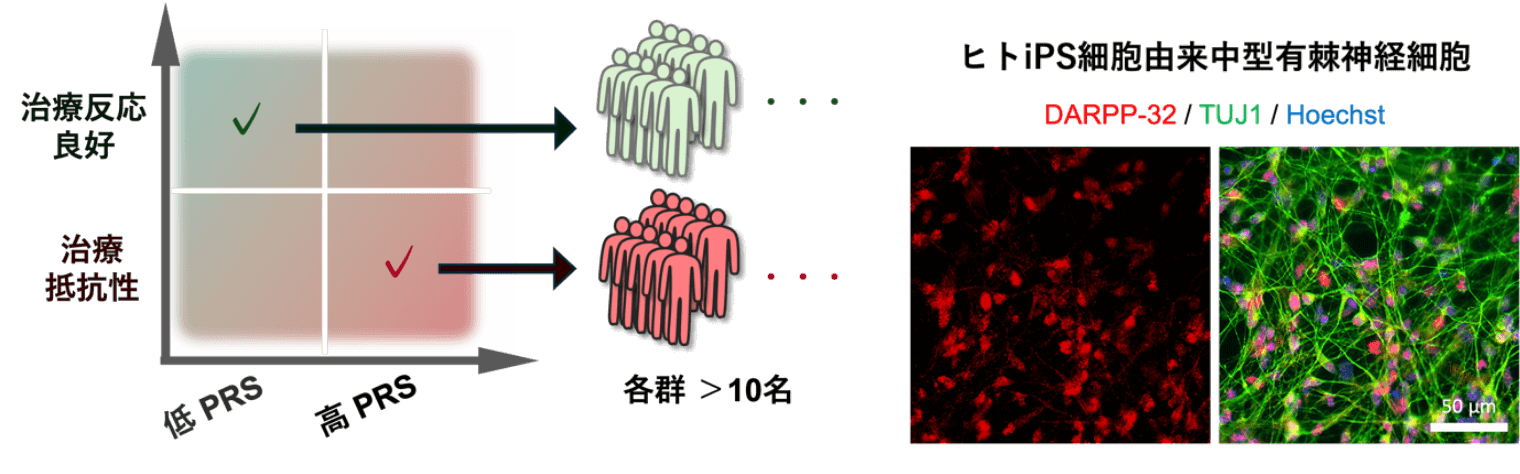 Figure 3. Establishment of iPS Cells Based on Stratification of Schizophrenia Patients and Development of a Novel Differentiation Method for Human iPS Cell–Derived Medium Spiny Neurons
Figure 3. Establishment of iPS Cells Based on Stratification of Schizophrenia Patients and Development of a Novel Differentiation Method for Human iPS Cell–Derived Medium Spiny Neurons
