New Biomarker for Immunoglobulin A Nephropathy Identified
Researchers find complement pathway proteins that could be treatment targets against the progression of IgA nephropathy
Immunoglobulin A (IgA) nephropathy results from the accumulation of IgA-containing immune complexes (IgA-ICs) and aberrant IgA antibodies in the glomerular mesangium. Now, researchers have found that complement pathway proteins are involved in the formation of these complexes. One such protein, complement factor H-related protein 1, is overexpressed in the glomeruli and IgA-ICs isolated from patients and could serve as a biomarker for disease susceptibility and progression, as well as a possible target for new therapies.
Immunoglobulin A (IgA) nephropathy is an autoimmune disease characterized by the deposition of circulating IgA-containing immune complexes (IgA-ICs) in the glomerular mesangium, leading to mesangial cell proliferation, enhanced extracellular matrix production, and variable infiltration of inflammatory cells. Because IgA nephropathy carries a lifelong risk of progression to end-stage kidney disease, optimal therapeutic strategies to halt renal deterioration are needed.
In accordance with the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines for IgA nephropathy and IgA vasculitis, therapeutic strategies are categorized into two groups: one targeting IgAN-specific drivers for nephron loss, and the other addressing the host’s genetic response to IgAN-induced nephron loss. The former is represented by immunosuppressive agents, whereas the latter mainly consists of comprehensive supportive care focused on managing cardiovascular and metabolic risk factors. However, systemic corticosteroid therapy has raised safety concerns and is therefore used only in carefully selected patients who can tolerate its adverse effects. Thus, unraveling the molecular pathogenesis of IgA nephropathy and identifying reliable biomarkers and actionable therapeutic targets are pivotal.
“The key IgA-IC-associated molecules and pathways involved in the development of glomerulonephritis remain unclear,” says Professor Kazuo Takahashi from the Department of Biomedical Molecular Sciences, Fujita Health University School of Medicine, Japan. To elucidate the molecular components of IgA-ICs and molecules involved in glomerular injury in patients with IgA nephropathy, a proteome study was conducted by a team of researchers led by Prof. Takahashi, along with Senior Assistant Professor Yukako Ohyama and Assistant Professor Yudai Tsuji from the same institute. The team performed proteomic analyses on stored remnant FFPE kidney tissues and on IgA-ICs isolated from patients with IgA nephropathy, as well as on samples obtained from controls. Their findings were made available online on November 26, 2025, and were published in volume 15 of the journal Scientific Reports on December 30, 2025.
The team found that complement pathway proteins, associated with both the classical and alternative, and terminal pathways were overexpressed in the glomeruli of patients with IgA nephropathy compared with normal kidney segments removed due to cancer. These included complement factor H-related protein (CFHR) 1, CFHR2, CFHR3, CFHR5, C1q chains B and C, and properdin. Serum CFHR1 levels and CFHR1 levels within circulating IgA-ICs were also significantly higher in patients than in healthy individuals and disease controls. Furthermore, colocalization of CFHR1 with mesangial IgA deposition was confirmed by double immunofluorescence staining of CFHR1 ad IgA using frozen kidney-biopsy sections.
Longitudinal studies showed that CFHR1 levels in circulating IgA-ICs were significantly reduced after two years of immunosuppressive treatment, whereas no significant change was observed in patients receiving supportive therapy. Interestingly, serum CFHR1 levels remained unchanged in both treatment groups.
What does this mean for the progression of IgA nephropathy? Prof. Takahashi speculates that elevated CFHR1 in circulating IgA-ICs may increase the likelihood of developing the disorder, adding, “Elevated circulating total CFHR1 levels in patients with IgA nephropathy may reflect activation of the alternative complement pathway upon exposure to mucosal microbial antigens bound to IgA.” CFHR1 may thus catalyze the formation of IgA-ICs around abnormal IgA molecules, which eventually get deposited in the kidney and cause inflammation.
These findings indicate that CFHR1 could serve as a new diagnostic marker for IgA nephropathy. “The quantification of CFHR1 within circulating IgA immune complexes holds promise as a novel diagnostic and prognostic biomarker for IgA nephropathy. Notably, several new therapeutic agents with distinct mechanisms of action—including complement-targeting drugs—are currently under development and clinical evaluation for IgA nephropathy. CFHR1 may also serve as a potential companion diagnostic marker to guide these emerging therapies,” remarks Prof. Takahashi. He adds that a better understanding of the mechanism of IgA-IC formation, including the roles played by complement pathway proteins, will lead to optimized patient management and better treatment outcomes.
Immunoglobulin A (IgA) nephropathy results from the accumulation of IgA-containing immune complexes (IgA-ICs) and aberrant IgA antibodies in the glomerular mesangium. Now, researchers have found that complement pathway proteins are involved in the formation of these complexes. One such protein, complement factor H-related protein 1, is overexpressed in the glomeruli and IgA-ICs isolated from patients and could serve as a biomarker for disease susceptibility and progression, as well as a possible target for new therapies.
Immunoglobulin A (IgA) nephropathy is an autoimmune disease characterized by the deposition of circulating IgA-containing immune complexes (IgA-ICs) in the glomerular mesangium, leading to mesangial cell proliferation, enhanced extracellular matrix production, and variable infiltration of inflammatory cells. Because IgA nephropathy carries a lifelong risk of progression to end-stage kidney disease, optimal therapeutic strategies to halt renal deterioration are needed.
In accordance with the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines for IgA nephropathy and IgA vasculitis, therapeutic strategies are categorized into two groups: one targeting IgAN-specific drivers for nephron loss, and the other addressing the host’s genetic response to IgAN-induced nephron loss. The former is represented by immunosuppressive agents, whereas the latter mainly consists of comprehensive supportive care focused on managing cardiovascular and metabolic risk factors. However, systemic corticosteroid therapy has raised safety concerns and is therefore used only in carefully selected patients who can tolerate its adverse effects. Thus, unraveling the molecular pathogenesis of IgA nephropathy and identifying reliable biomarkers and actionable therapeutic targets are pivotal.
“The key IgA-IC-associated molecules and pathways involved in the development of glomerulonephritis remain unclear,” says Professor Kazuo Takahashi from the Department of Biomedical Molecular Sciences, Fujita Health University School of Medicine, Japan. To elucidate the molecular components of IgA-ICs and molecules involved in glomerular injury in patients with IgA nephropathy, a proteome study was conducted by a team of researchers led by Prof. Takahashi, along with Senior Assistant Professor Yukako Ohyama and Assistant Professor Yudai Tsuji from the same institute. The team performed proteomic analyses on stored remnant FFPE kidney tissues and on IgA-ICs isolated from patients with IgA nephropathy, as well as on samples obtained from controls. Their findings were made available online on November 26, 2025, and were published in volume 15 of the journal Scientific Reports on December 30, 2025.
The team found that complement pathway proteins, associated with both the classical and alternative, and terminal pathways were overexpressed in the glomeruli of patients with IgA nephropathy compared with normal kidney segments removed due to cancer. These included complement factor H-related protein (CFHR) 1, CFHR2, CFHR3, CFHR5, C1q chains B and C, and properdin. Serum CFHR1 levels and CFHR1 levels within circulating IgA-ICs were also significantly higher in patients than in healthy individuals and disease controls. Furthermore, colocalization of CFHR1 with mesangial IgA deposition was confirmed by double immunofluorescence staining of CFHR1 ad IgA using frozen kidney-biopsy sections.
Longitudinal studies showed that CFHR1 levels in circulating IgA-ICs were significantly reduced after two years of immunosuppressive treatment, whereas no significant change was observed in patients receiving supportive therapy. Interestingly, serum CFHR1 levels remained unchanged in both treatment groups.
What does this mean for the progression of IgA nephropathy? Prof. Takahashi speculates that elevated CFHR1 in circulating IgA-ICs may increase the likelihood of developing the disorder, adding, “Elevated circulating total CFHR1 levels in patients with IgA nephropathy may reflect activation of the alternative complement pathway upon exposure to mucosal microbial antigens bound to IgA.” CFHR1 may thus catalyze the formation of IgA-ICs around abnormal IgA molecules, which eventually get deposited in the kidney and cause inflammation.
These findings indicate that CFHR1 could serve as a new diagnostic marker for IgA nephropathy. “The quantification of CFHR1 within circulating IgA immune complexes holds promise as a novel diagnostic and prognostic biomarker for IgA nephropathy. Notably, several new therapeutic agents with distinct mechanisms of action—including complement-targeting drugs—are currently under development and clinical evaluation for IgA nephropathy. CFHR1 may also serve as a potential companion diagnostic marker to guide these emerging therapies,” remarks Prof. Takahashi. He adds that a better understanding of the mechanism of IgA-IC formation, including the roles played by complement pathway proteins, will lead to optimized patient management and better treatment outcomes.
Reference
Title of original paper:
Complement proteins associated with circulatory and glomerular IgA-containing immune complexes in patients with IgA nephropathyJournal:
Scientific ReportsDOI:
10.1038/s41598-025-29024-zAbout Professor Kazuo Takahashi from Fujita Health University
Dr. Kazuo Takahashi is a Professor at the Department of Biomedical Molecular Sciences at Fujita Health University School of Medicine, Japan. His research focuses on the proteomics of renal disorders. Prof. Takahashi has authored over 130 papers and book chapters in these fields. He is a member of several prestigious medical associations, including the American Society of Nephrology, the Japanese Society of Nephrology, the Japanese Society for Dialysis Therapy, and the Japanese Society of Anatomy. In addition to his research and academic work, Prof. Takahashi serves as a nephrologist at Fujita Health University Hospital.
Funding information
- Oriental Yeast CO., LTD.
- Japan Society for the Promotion of Science KAKENHI (Grant numbers: 20K08623, 20K22915, 22K08366, 22K16253, and 23K07729)
- Hori Sciences and Arts Foundation
- Yukiko Ishibashi Foundation
- Aichi Jinzou Foundation
- Jan Novak is supported in part by UAB research-acceleration funds and a gift from IGA Nephropathy Foundation
Image
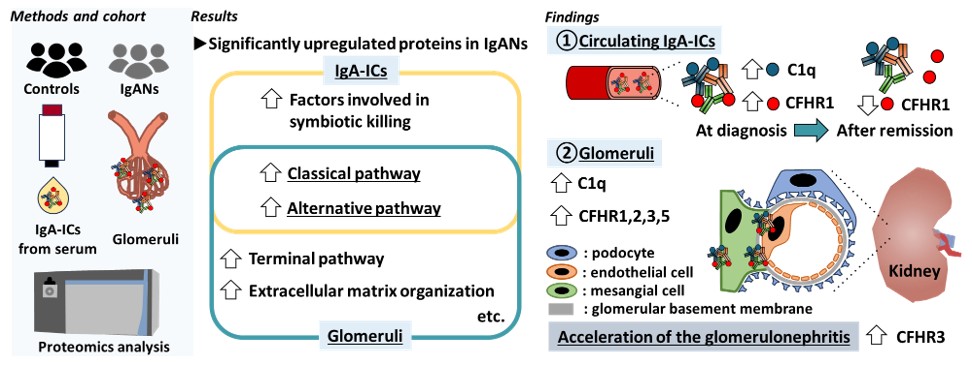
Image title: CFHR1 is involved in IgA immune complex formation
Image caption: Complement pathway proteins are involved in immunoglobulin A-containing immune complex (IgA-IC) formation and deposition in the kidneys. Elevated complement factor H-related protein 1 (CFHR1) may be a biomarker for susceptibility to IgA nephropathy. Immunosuppressive treatment reduces IgA-IC formation and increases CFHR3 in kidney tissue.
Image credit: Professor Kazuo Takahashi from Fujita Health University School of Medicine, Japan
License type: Original content
Usage restrictions: Cannot be reused without permission.


